Patterns of life on Scotland’s rocky seashores are shaped by differences in waves, nutrients and water temperature around the coast. Responses to climate and other changes reflect these patterns. Alongside recent increases in seaweeds and declines of mussels, warm-water species have increased due to climate warming since the 1980s. This assessment is based on extensive surveys of most of Scotland’s rocky coasts in 2014/15 and will form the basis for detecting change in future assessments.
Introduction
The state of ecological communities on Scotland’s coasts is best understood through an examination of the primary influences on the distribution and abundance of the main species living there, namely waves, light, nutrients and temperature. Here, the main types of communities and their arrangement is reviewed, and the recent response of life on rocky shores to climate warming is interpreted through the lens of sensitivity of different species to temperature. Other major changes have occurred over the last decade, notably the decline of blue mussels and increase in seaweeds, but their causes remain unknown.
Habitat and ecological community types
The arrangements of types of rocky shore communities along environmental gradients such as wave exposure and sea temperature are strong indicators of likely community responses to changes in those factors over time. With future warming, for example, the composition of rocky shore communities is expected to become more like that seen in currently warmer conditions to the south and west of Scotland.
Wave exposure
Exposure of coasts to waves is the main factor determining both the habitat type and the make-up of the communities of animals and plants on rocky shores. Actual measurements of size of waves and frequency of extreme wave events is a time-consuming and costly activity. Instead, wave exposure is commonly estimated using a measure of how much the shoreline faces the open sea, expressed by the distance to the nearest land (wave “fetch”) in all directions around a coastal location (Burrows et al., 2008; Burrows, 2012).
Scotland has a broad mix of seashores across the categories of wave exposure (Figure 1). Wave-exposed shores are found all around the coast, with the most exposed shores on the outer coasts of the Hebrides (Islay, Tiree, Barra, the Uists and Lewis), the North Coast, Orkney and Shetland. East coast shores have similar wave fetch but do not face the Atlantic swells of the west coast
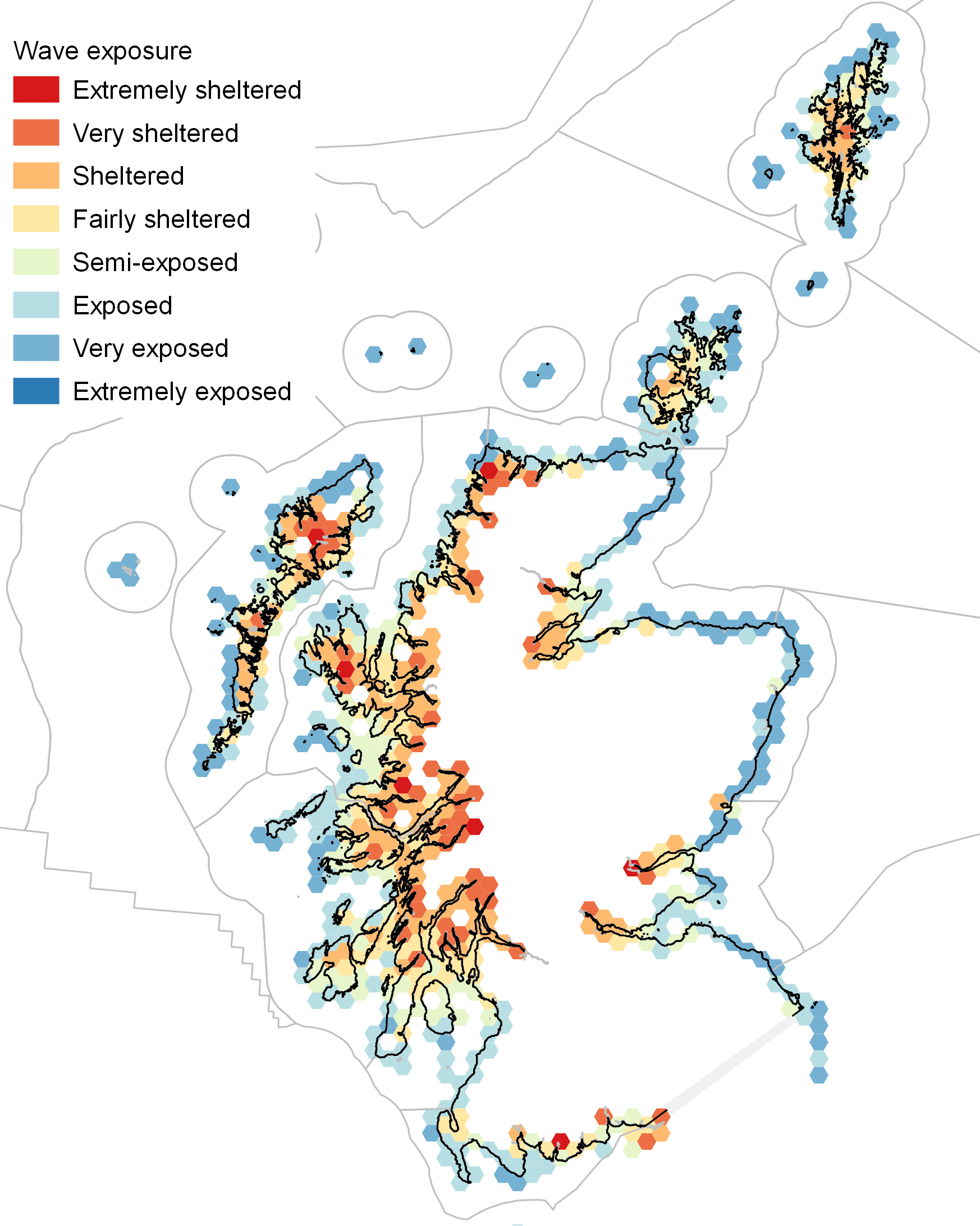
Wave-sheltered shores predominate in the sea lochs of the west coast, and in the firths and voes of the Hebrides, Orkney and Shetland. Wave shelter on the east coast is limited to the shores of the Dornoch, Cromarty and Moray Firths in the north and Firths of Tay and Forth in the south.
Habitat types and wave exposure
Out of an estimated total length of 9,000 km for Scotland’s coastline (estimated by a box-counting method using 200 m-sized grid cells, about 1:250,000 scale), about 54% (4,800 km) is composed of rock (Defra Intertidal Substratum dataset) with a further 11% (1,010 km) of rock mixed with other sediment types. The remaining coast is made up of sand (14%, 1,240 km), shingle (14%, 1,280 km), and mud (4%, 330 km) or a mixture of sediments (mud, sand, shingle, 4%, 330 km). Man-made structures make up about 50 km (0.6%).
The proportion of each of these seashore types changes with wave exposure (Figure 2). Man-made and mud shores are only found in sheltered conditions, while rock forms an increasing proportion of the coastline towards more wave-exposed conditions (Figure 2 right). Rock mixed with shingle forms nearly half of the rocky coastline in sheltered areas but the proportion of coastline with rock and smaller mobile rock material (shingle) declines with increasing wave exposure.

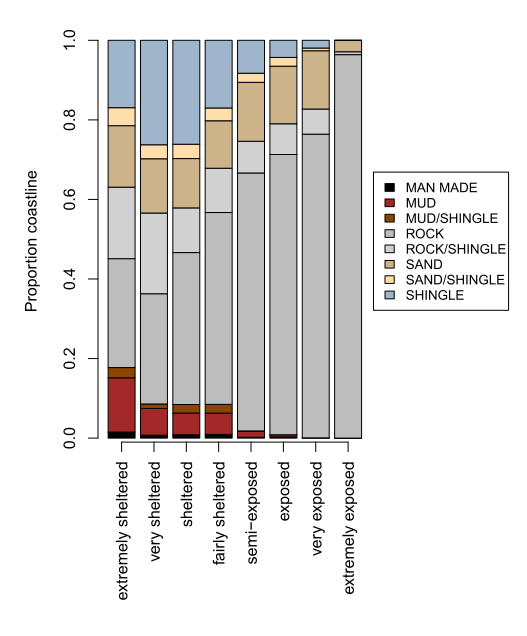
Figure 2: (left) Length of Scotland’s coastline of different geological types across wave-exposure categories. (right) Proportion of each shore type changes with wave exposure.
Ecological communities and wave exposure
The communities of animals and plants living on rocky seashores change markedly across the wave exposure gradient. In very wave-exposed conditions, red algae can dominate the mid to lower shore, often forming beds of pink turf-forming coralline algae (Corallina officinalis, Figure 3) or the red seaweed dulse (Palmaria palmata, Figure 4). In less energetic, but still exposed conditions, shores are mostly dominated by filter-feeding barnacles and mussels (Figure 5) with wave-tolerant forms of algae species more commonly found in more sheltered places, such as the bladderless form of bladder wrack (Fucus vesiculosus evesiculosus).


Figure 4: A very wave-exposed shore dominated by dulse (the red alga Palmaria palmata), Orkney Mainland, Banks. © Mike Burrows, SAMS.

The complexity of rock formations in wave exposed areas can provide refuges for less wave-tolerant species, meaning that a greater mix of species can be found on more topographically complex shores. Similarly, shores with more horizontal surfaces tend to have ecological characteristics more typical of less wave exposed localities, as waves breaking across such reefs at high tide may lose energy towards the landward side of the shore.
Large brown seaweeds cover an increasing proportion of shores when progressing from semi-exposed towards wave-sheltered conditions (Figure 6 to Figure 10).

Figure 6: A semi-exposed shore with upper-shore barnacles and lower-shore algae (Fucus serratus) and exposed kelp (Laminaria digitata), Glengorm, Mull. © Mike Burrows, SAMS.




Seaweeds including bladder wrack (Fucus vesiculosus) and serrated wrack (Fucus serratus) dominate shores in conditions of intermediate wave exposure (Figure 7, Figure 8), while the most wave-sheltered shores can be almost entirely covered by egg wrack (Ascophyllum nodosum, also known as rockweed, Figure 10). Egg wrack plants can be up to 1.5 m long and can form the highest biomass of any seaweed on the shore, typically up to 10 kg of wet mass per metre squared. Commercial harvesting of intertidal seaweed is largely focussed on egg wrack, with significant harvesting activity in North Uist and Lewis (Burrows et al., 2010).
Light and temperature as influences on community composition
Light is essential for seaweed and the clarity of water may be important for the growth of seaweed, even if exposed to air during low tide. Satellite data on the colour of the ocean surface (Figure 11 right) shows that high concentrations of phytoplankton cells are often found in the waters around urban centres including the Firths of Clyde and Forth, where nutrients promote plant growth. Suspended sediment particles in the water also reduce light penetration (Figure 11 left), with high levels of suspended material in the Solway Firth and across the east coast.
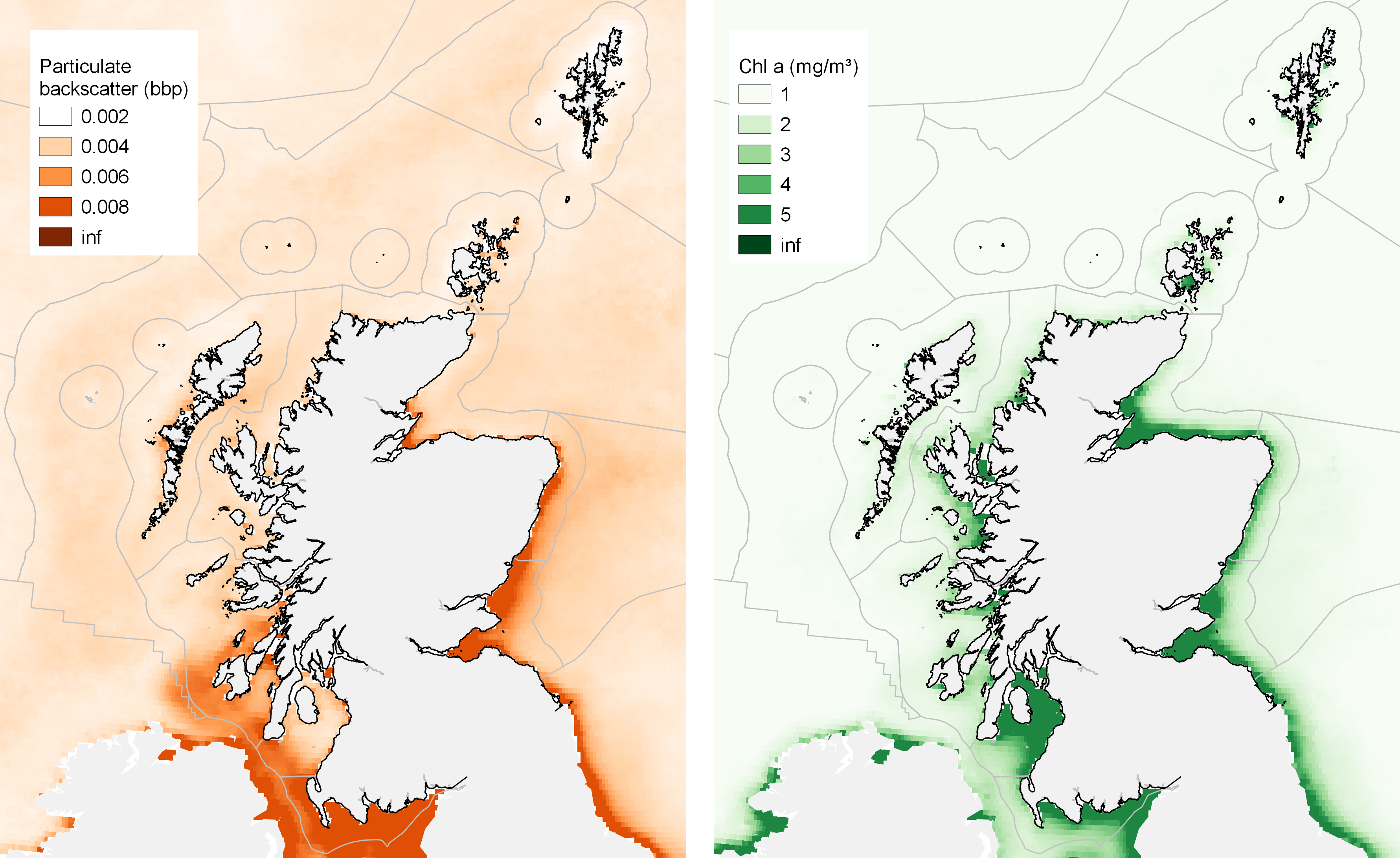
The number of species of seaweed found on the shore is reduced in areas with reduced light penetration (Burrows et al., 2014). Whether the limits imposed by the light available inhibits seaweed growth in such places or, alternatively, the increased amount of phytoplankton and other suspended material promotes the growth of filter-feeding animals (primarily barnacles and mussels) that may compete for space is not yet known. The switch between seaweed-dominated communities and filter-feeder-dominated communities may be a useful reflection of coastal water quality. The regional differences in rocky shore communities between areas of low and high light levels suggest likely responses to changing water quality in any given area, with increased diversity of seaweeds expected with improving quality. In contrast, increased coastal nutrients, for example, is likely to lead to greater concentrations of phytoplankton and a consequent shift in dominance of rocky shore communities towards barnacles and mussels.
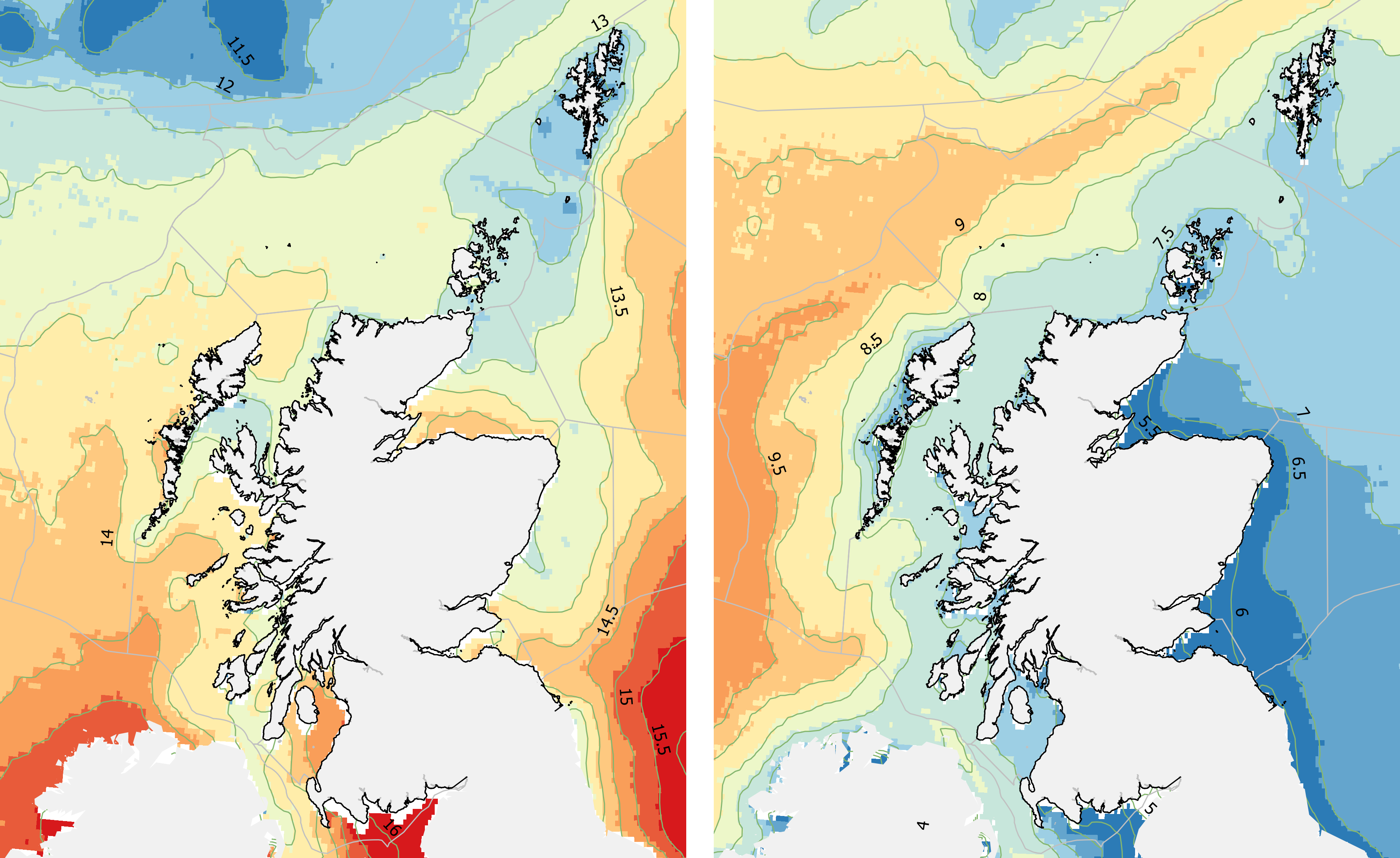
Sea temperature is important for rocky shore communities, mostly through the effect on those species of animals and plants that are at either the southern or northern limits of their distribution in Scotland. Such species are generally not large or habitat-forming but their presence is associated with cooler or warmer water temperatures, while fluctuations or trends in the abundance or prevalence of warm-water or cold-water species are taken as strong indications of a response to climate change (see Climate change section).
Summer sea temperatures (Figure 12 left) tend to be cooler in the north while winter sea surface temperatures, such as those in February (Figure 12 right), are cooler in the east. Southern, warm-water species on rocky coasts are generally restricted to the west coast of Scotland and the Northern Isles, and are therefore better aligned with the pattern of winter rather than summer temperatures.
Species distribution
Broad patterns of species distributions and composition of rocky shore communities across Scotland are well known. Earlier studies established the trends for species (e.g. barnacles, Moore et al., 1939; Crisp et al., 1981; wracks, Walker, 1947; other algae Hardy et al., 2006) and for whole communities (Lewis & Powell, 1960; Lewis 1964). Extensive efforts in the 1970s, 80s and 90s produced detailed inventories of species presence on these shores (BODC web site), and form the bulk of current databases (e.g. JNCC Marine Recorder) of rocky coastal marine conservation features essential for coastal planning.
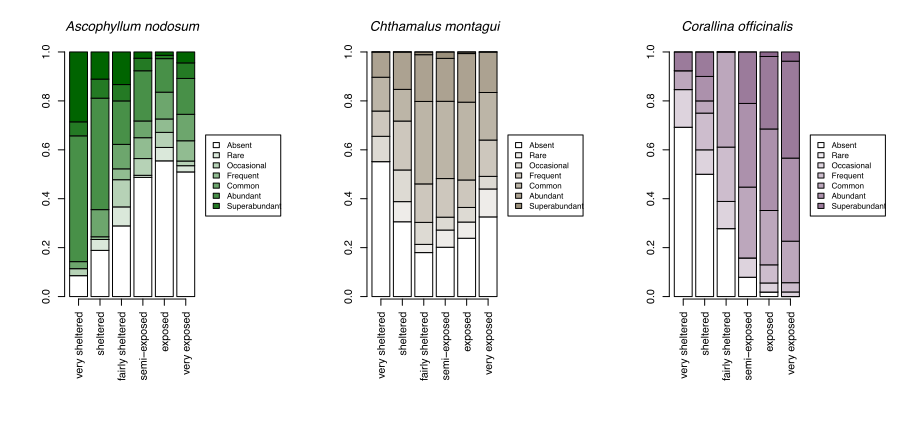
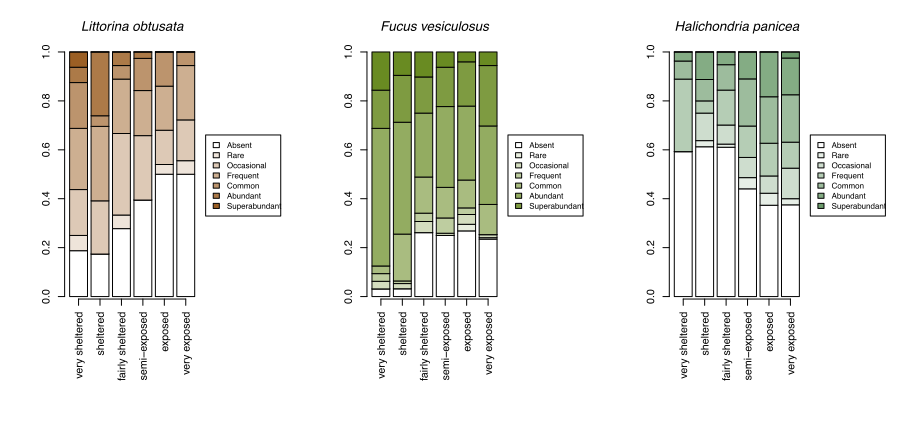
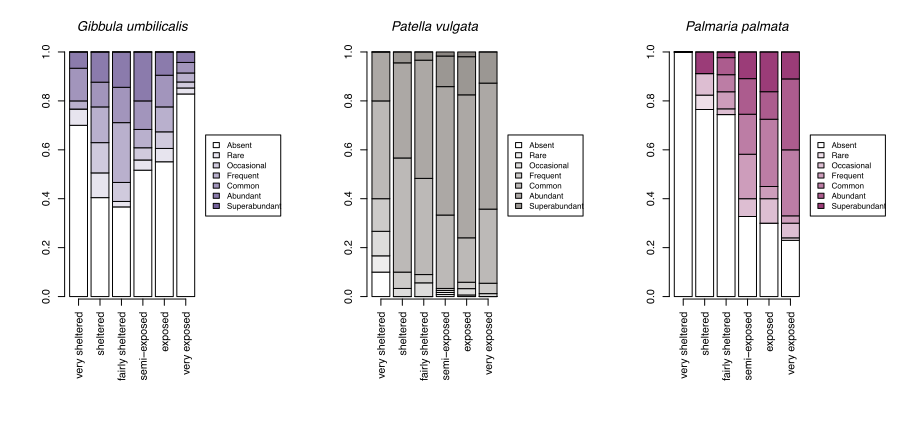
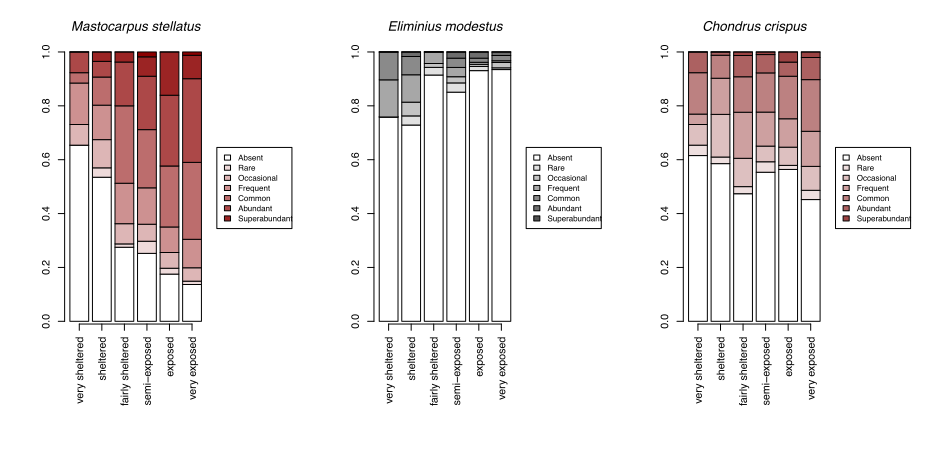
With growing concerns about the impacts of climate change, systematic surveys of animals and plants on rocky shores all around Scotland have been made more frequently since 2000 with the aim of detecting ecological change as it happens and establishing baselines for future change. These most recent records show patterns of changing abundance for each species across wave exposure gradients (Figure 13).
 |
 |
 |
 |
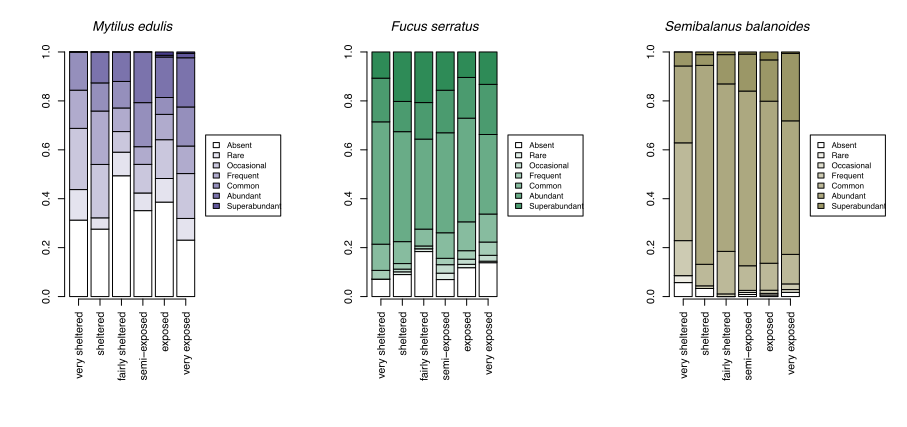 |
Changes in averages are less informative than the changes in the spread of sites where each species reaches a particular abundance category. For egg wrack (Ascophyllum nodosum), the probability of the species being super abundant (>60% cover) increases towards wave shelter, with the probability of it being completely absent from a shore increasing towards wave exposure. Red seaweeds, including dulse (Palmaria palmata, also Figure 4), false Irish moss (Mastocarpus stellatus) and coral weed (Corallina officinalis, also Figure 3) show the opposite pattern, becoming progressively more likely to be in higher abundance categories towards wave exposed conditions.
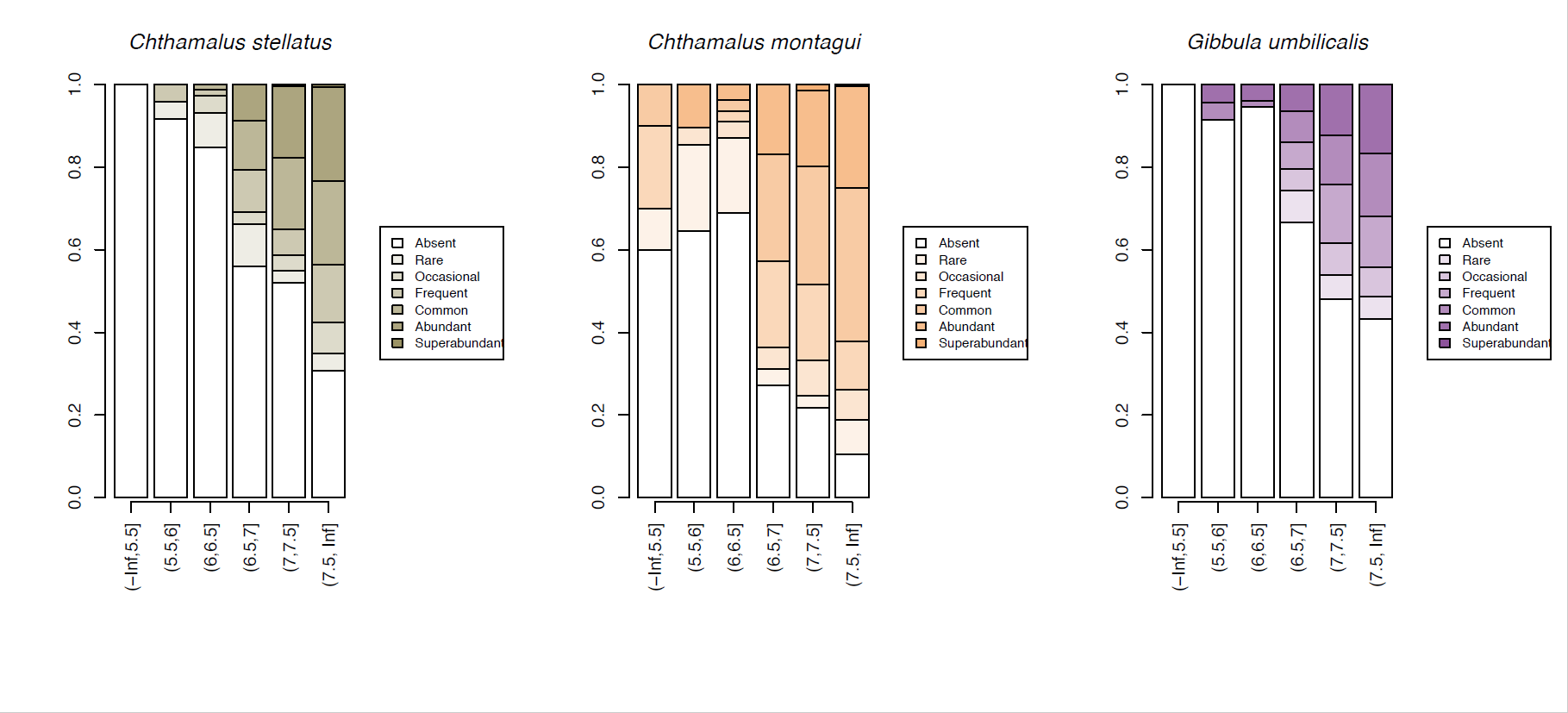
A few species, such as the warm-water acorn barnacles Chthamalus montagui and Chthamalus stellatus and the flat topshell Steromphala (previously Gibbula) umbilicalis are restricted to west and north coast seashores, and are thereby more likely to be abundant in areas of warmer February temperatures (Figure 14).
Climate change
Records of sea temperatures show a consistent increase in average, maximum and minimum temperatures around Scotland over the last 150 years (Figure 15), with the most pronounced changes happening since 1990, with a nearly 1°C rise in summer temperatures, a 0.5°C increase in average temperatures, and a smaller increase in winter minimum temperatures.
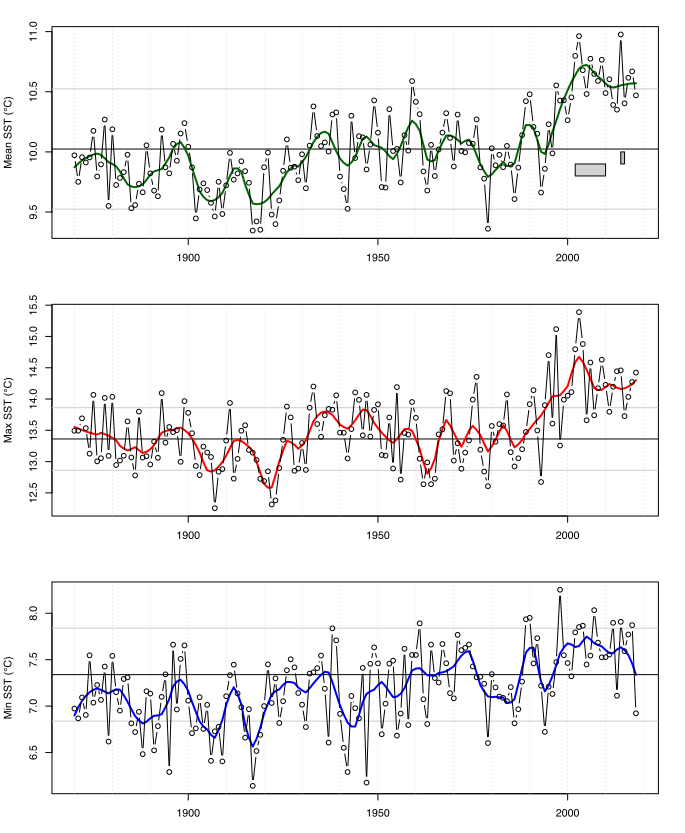
Figure 15: (top) Annual mean sea surface temperature for northern UK waters (Hadley Centre HadISST v1.1 monthly 1-degree dataset, 8°W-1°E 54-61°N), (mid) maximum temperature, (bottom) minimum temperature. Horizontal lines show 1960-1991 averages +/- 0.5°C. Shaded boxes in the top plot show survey periods.
Responses of ecological communities to the currently warming climate are primarily driven by the temperature sensitivities of their component species of animals and plants, as revealed by their geographical distributions. Rocky shore communities in Scotland are no exception to this rule (Burrows, 2017).
Widespread systematic monitoring of UK rocky shores (including Scotland and Ireland) began in 2001 with the aim of detecting responses to climate warming (among other stressors) (Mieszkowska et al., 2005). This programme started immediately after two decades of rapid warming and showed expansions by the early 2000s in the distribution of warm-water species (Mieszkowska et al., 2006) since the mid-1980s that were consistent with warming, notably an apparent expansion of the flat topshell Steromphala (previously Gibbula) umbilicalis along the Caithness coast.
Despite a more intensive survey effort in 2014/15, including a visit to Shetland not surveyed in the previous decade, there was not strong evidence for range shifts or changes in abundance that could be easily attributed to temperature. Some warm-water forms such as Poli’s stellate barnacle (Chthamalus stellatus) (Figure 16) were found in low numbers in places outside their historical ranges in 2014/15 but only after targeted searching of shores with the right kinds of conditions suitable for those particular species. Since little temperature change had occurred since the previous survey (Figure 15), this lack of significant climate-driven change was not unexpected.
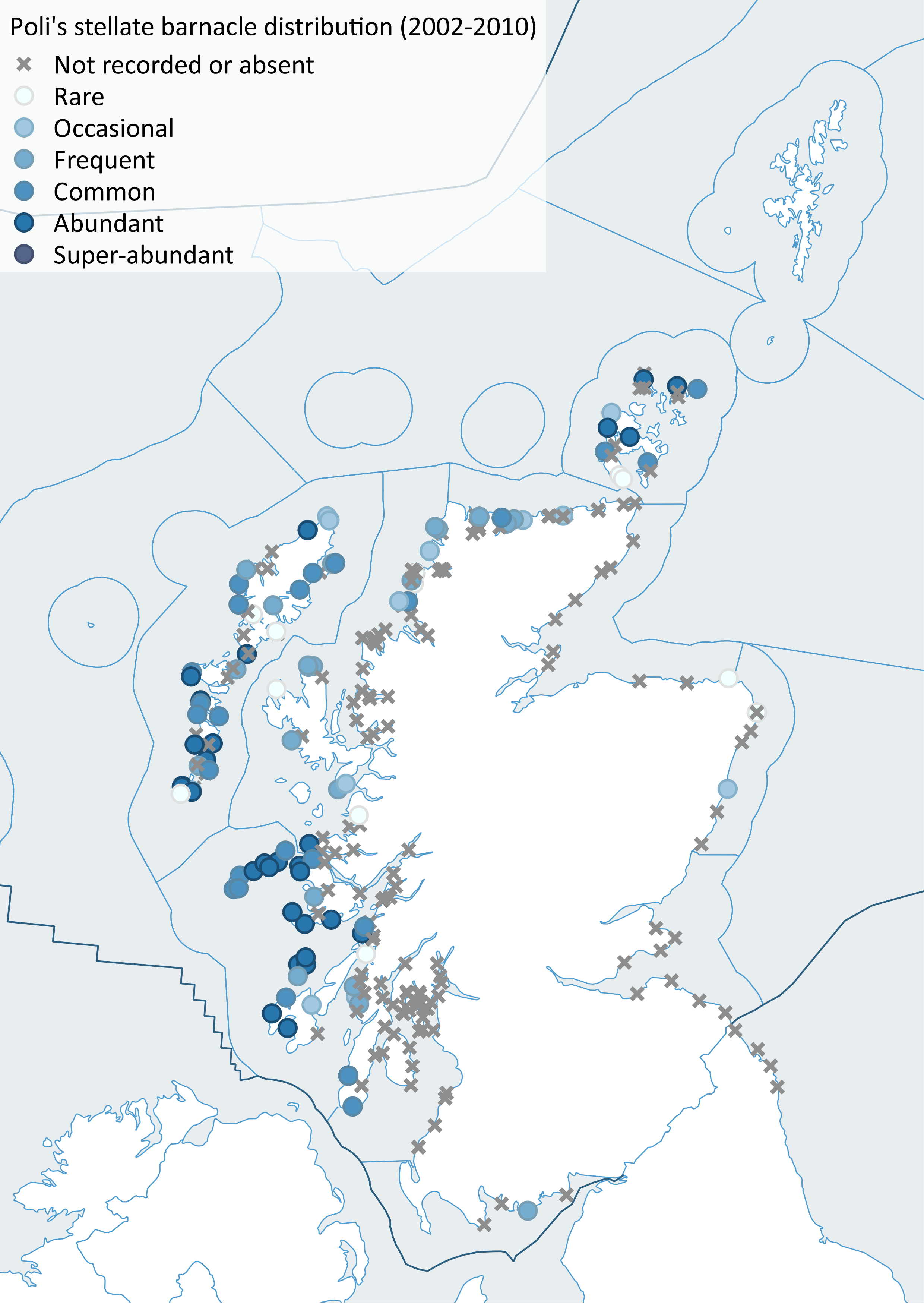
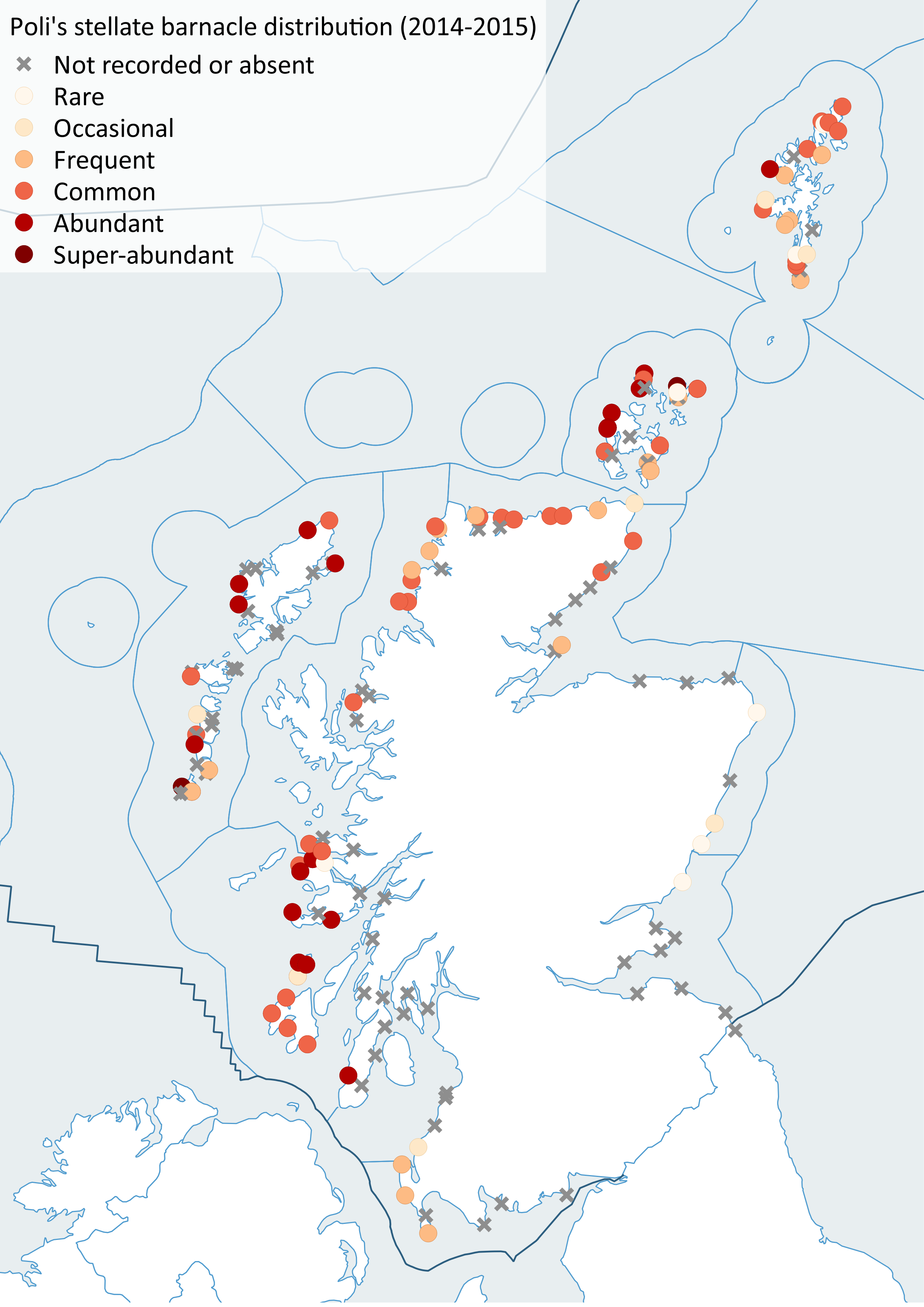
Figure 16: Distribution of Poli's stellate barnacle (Chthamalus stellatus), left (2002-2010), right (2014-2015).
While dramatic changes are difficult to demonstrate, it has been possible to show how changes across many species considered together over a longer period are happening in relation to warming. Warm-water species, those from places warmer than prevailing conditions in Scotland whose distributions may extend into southern Europe and African coasts, tend to increase in abundance with warming temperatures while those from colder places, here from Scotland towards the Arctic, tend to decline with warming. One method of expressing this change across multiple species is to estimate the average across all species in a community of the geographical midpoint temperatures across their distribution range ((Burrows et al., 2017). Termed the Community Temperature Index, this measure directly reflects the changing dominance of cold- and warm-water species in relation to temperature change.
The best example of the shift from cold-water to warm-water species is from the environmental monitoring programme around the oil terminal in Sullom Voe, Shetland. Rocky shores in the area have been surveyed since before the construction of the terminal in 1976. Average sea temperatures in the area have increased from around 9.5°C in the early 1980s to around 10.2°C in the late 2010s. . After a steep decline following a cold spell in 1979, the average species midpoint temperature (the Community Temperature Index) for the community has increased over the same period by a smaller quantity (Figure 17), but nonetheless in line with the temperature increases. Seaweed species did not show this shift, largely because seaweeds in the area have generally similar temperature tolerances and have therefore not changed in their abundance relative to each other.
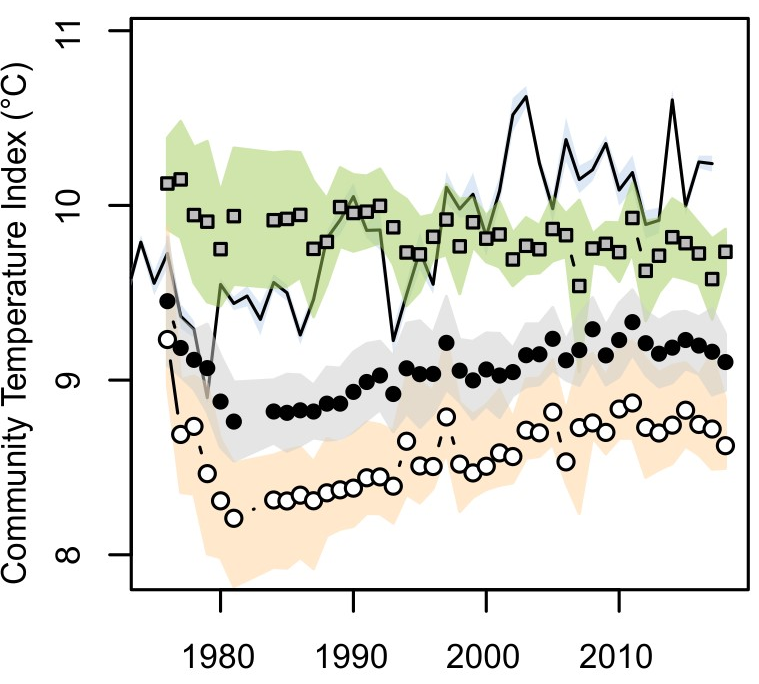
Figure 17: Changes in Community Temperature Index (CTI) for rocky shores around Sullom Voe, Shetland since 1976. Annual CTI values for all species in the community (•) and for animal species (o) shows a steady increase since 1980, while seaweed species composition remains stable (). Coloured ribbons show variability among sites. The solid lines shows average annual sea surface temperatures in the area.
Other recent changes
While changes between the early 2000s and 2014/2015 were not immediately attributable to climate change, largely because of the lack of a temperature increase over the period, there were notable increases and declines in species between the two periods (Figure 18). The biggest decline was in blue mussels (Mytilus edulis), showing reduced abundance at over 50% of the 154 sites revisited.
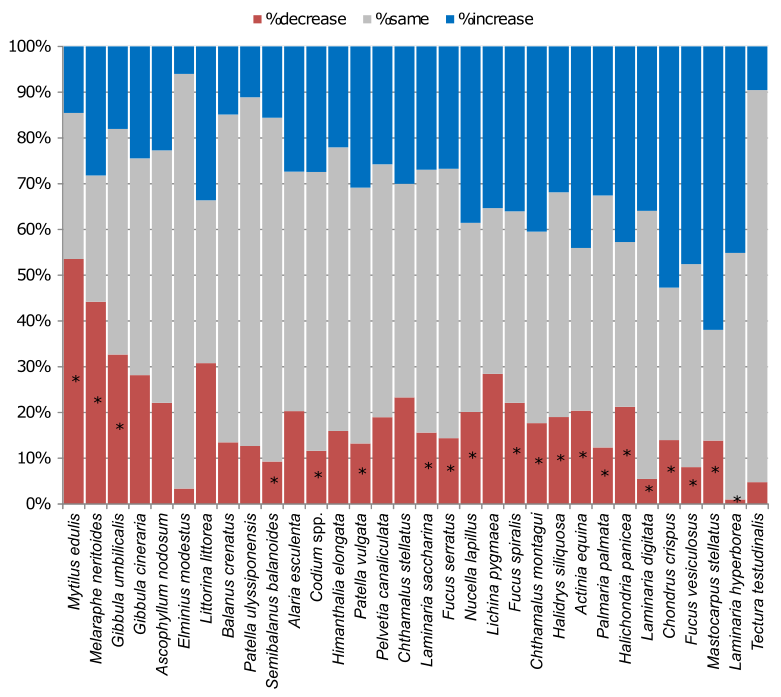
Many other species, mostly seaweeds, showed widespread increases over the same period. While the causes of these changes remain unknown, a possible candidate could be ocean acidification, since declining pH in coastal waters has been associated with declines in mussels in the Pacific coast of North America (Wootton et al., 2008), while in laboratory experiments algae show increases in performance in the lowered pH associated with more dissolved CO2 (Kroeker et al., 2013). Whether such changes are part of a longer-term trend or reflect short-term variations in the success of the species remains to be seen.
Scotland has an extensive and varied coastline comprising approximately 50% rocky and 50% sedimentary intertidal habitat. Large stretches of the Mainland west coast and Northern Isles are predominantly rocky whereas on the west coast of the Outer Hebrides and the Mainland east coast it is much more patchy with rocky shores and cliffs interspersed by large stretches of sandy and muddy coastline. Intertidal habitats are affected by numerous physical variables including wave exposure, salinity, temperature and tides which dictate what animals and plants are found on specific shores. The subtidal communities are strongly affected by factors such as the availability of light, wave action, tidal stream strength and salinity. Rocky shallow continental shelf habitats are typically dominated by seaweeds and in deeper areas below the photic zone (about 50 m) communities comprise exclusively animals. Shallow subtidal sediments in places support habitats such as seagrass beds and maerl, a red seaweed with a hard chalky skeleton that forms small twig-like nodules which accumulate to form loosely interlocking beds, creating the ideal habitat for a diverse community of organisms. Typically sedimentary habitats are dominated by a range of burrowing animal species.
The Biogenic habitats assessment catalogues the loss in extent of six biogenic habitats (all Priority Marine Features): blue mussel, horse mussel, flame shell, maerl, seagrass beds, and serpulid aggregations. The Predicted extent of physical disturbance to seafloor assessment uses the degree of exposure to demersal fishing activity as a proxy for habitat condition. The Intertidal seagrass assessment is a first attempt to understand the ecological health of Scottish intertidal seagrass and is restricted to six sites.
The Case study: Biogenic habitat enhancement highlights the efforts now being made to enhance the status of some biogenic habitats through activities aimed at aiding their recovery and restoration. In other cases, where damage to Priority Marine Features has occurred, positive action is taken as demonstrated in the Case study: Protecting the Loch Carron flame shell beds where emergency measures were put in place to prevent further damage and subsequently a Marine protected Area was designated. The fact the prevention is better than cure is illustrated by the Case study: Persistent damage to the Loch Creran serpulid reefs where damage that was first observed in 1998 still shows little evidence of recovery. The value of long-term monitoring of specific sites is illustrated by the Case study: Intertidal rock which highlights how looking at changes at a community scale helps separate natural variations on species abundances from longer term community trends. The growing awareness of natural capital and ecosystem services provided by the marine environment is illustrated by the three case studies Case study: Blue carbon in Scottish maerl beds; Case study: Blue carbon in Scottish marine sedimentary environments; Case study: Blue carbon: the contribution from seaweed detritus which highlight the importance of marine habitats in climate change mitigation through the capture and storage of blue carbon, and the need for the protection of such habitats from various anthropogenic activities. Despite a long history of intertidal and subtidal survey work there remains significant gaps in knowledge. The Case study: Seabed habitats in territorial waters - the evolving knowledge-base charts the ongoing surveys that have been undertaken by government agencies and citizen science initiatives to further expand and improve the knowledge base.


